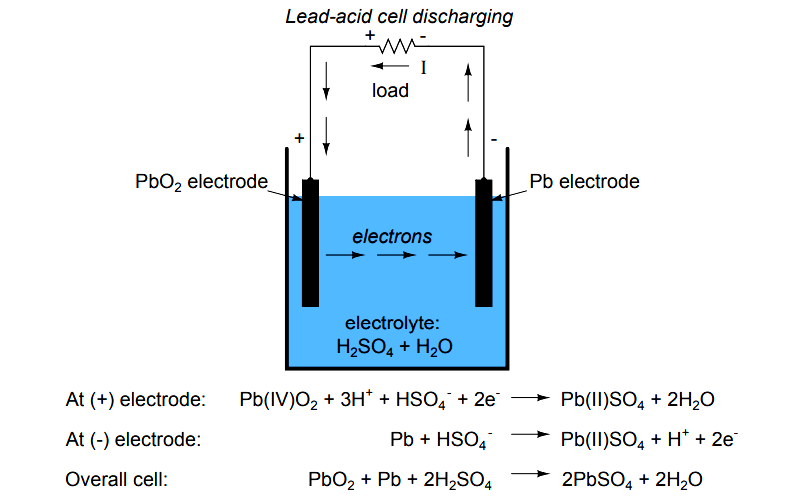
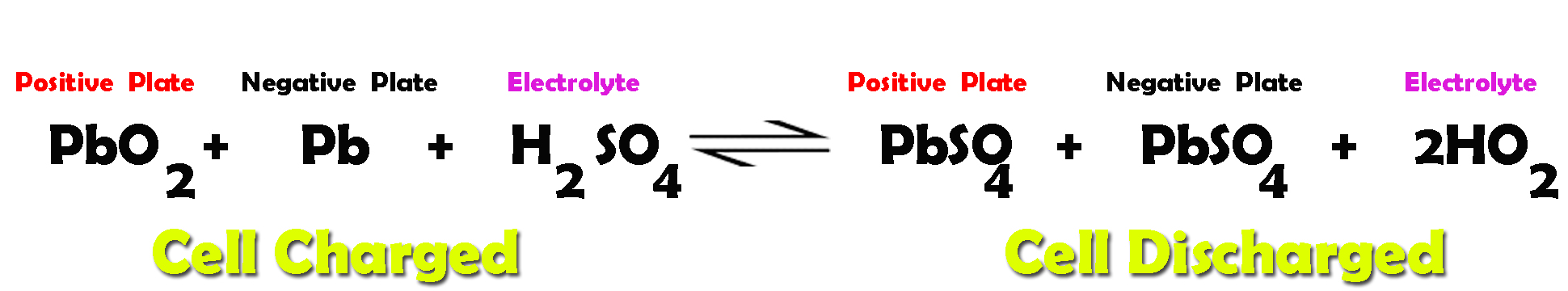
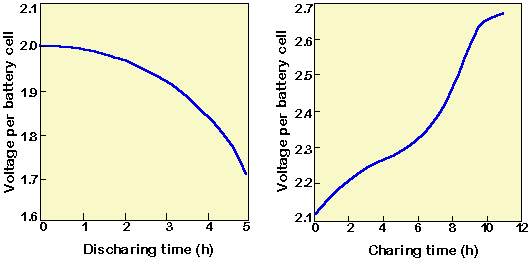
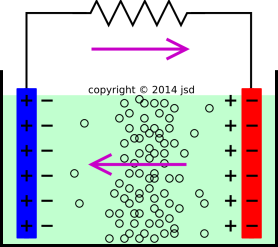
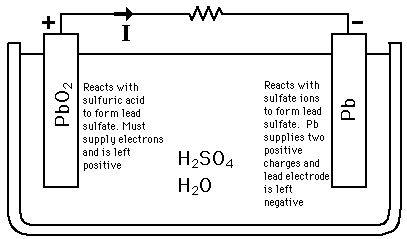
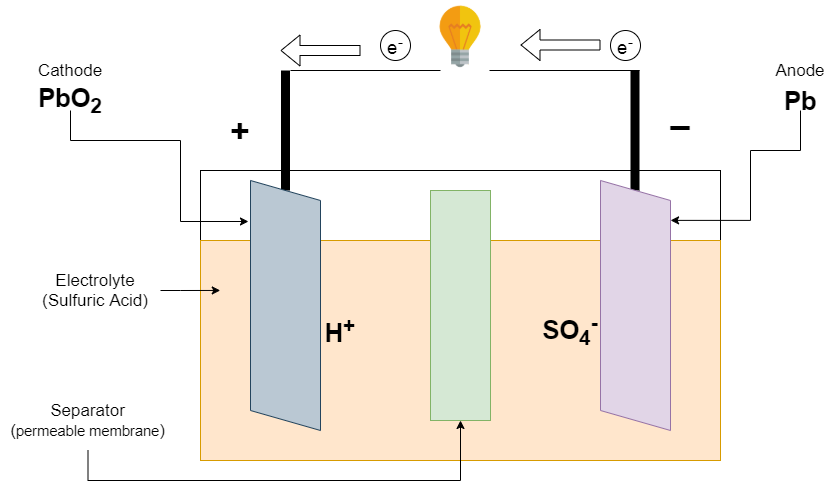
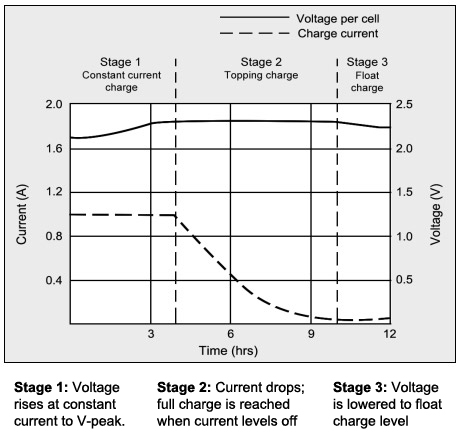
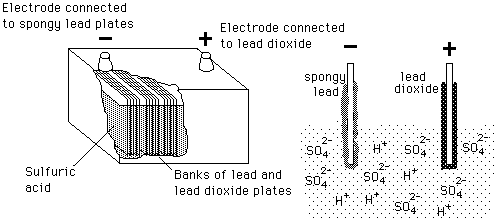
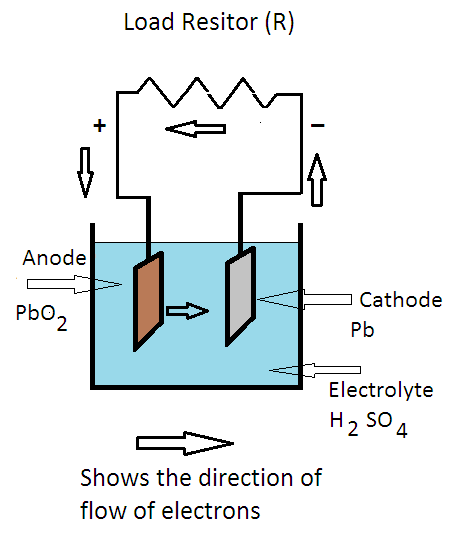
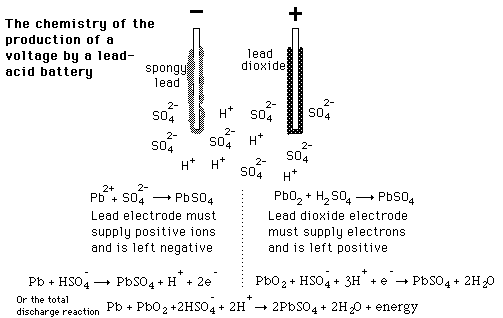SOLVED: The electrochemical reaction for a SLI lead-acid battery is: Pb(s) + PbOz(s) + 2HzSO4(aq) 2PbSO4(s) + 2HzO() EO2.041V Write down the Nernst Equation of above reaction marks) Explain why this Nernst

The half reactions that occur in a lead acid battery are: `PbSO_(3)(s)+2e^(-)toPb(s)+SO_(4) - YouTube

Discharge and charge reactions at the negative plate of a leadeacid cell. | Download Scientific Diagram